Stem cells are undifferentiated cells that have the capacity to proliferate in undifferentiated cells both in vitro and in vivo (self-renewal) and to differentiate into mature specialized cells. However, in some clinical trials serious adverse events have been reported, which emphasizes the need for additional knowledge, particularly with regard to biological mechanisms and long term safety. In contrast, the vast majority of small-sized clinical trials conducted with mesenchymal stem/stromal cells (MSC) in regenerative medicine applications has not reported major health concerns, suggesting that MSC therapies could be relatively safe. risk on tumour formation) are considered high, if not perceived to be unacceptable. Based on their characteristics of unlimited self-renewal and high proliferation rate the risks associated with a product containing these cells (e.g. risks observed in animal studies) include tumour formation, unwanted immune responses and the transmission of adventitious agents.Ĭurrently, there is no clinical experience with pluripotent stem cells (i.e. risks identified in clinical experience) or potential/theoretical risks ( i.e. Together these factors determine the risk profile associated with a stem cell based medicinal product. The risk profile of stem cell based medicinal products depends on many risk factors, which include the type of stem cells, their differentiation status and proliferation capacity, the route of administration, the intended location, in vitro culture and/or other manipulation steps, irreversibility of treatment, need/possibility for concurrent tissue regeneration in case of irreversible tissue loss, and long-term survival of engrafted cells. Evaluation of potential risks should be a prerequisite step before clinical use of stem cell based medicinal products. These risks deserve a thorough discussion within the perspective of current scientific knowledge and experience. Despite the clinical potential of stem cell based medicinal products there are also potential and unanticipated risks. The potential of stem cell therapies has long been recognised and the creation of induced pluripotent stem cells (iPSC) has boosted the stem cell field leading to increasing development and scientific knowledge. 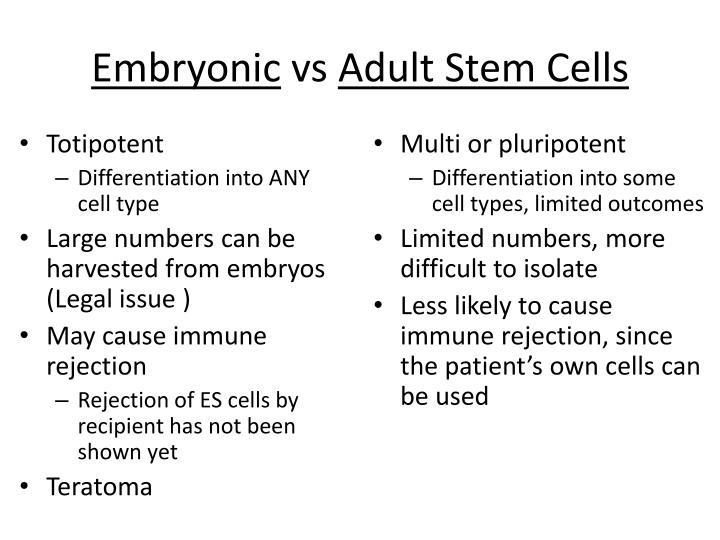
Patients could be exploited by paying for expensive treatments and being given false hope of a cure, as stem cell therapies are only in their developmental stages.Stem cell therapy holds the promise to treat degenerative diseases, cancer and repair of damaged tissues for which there are currently no or limited therapeutic options. Much of the research is being carried out by commercial clinics, so reported successes are not subject to peer review.Whether the benefits of stem cell use outweigh the objections.The importance of educating the public about what stem cells can and can't do.At what stage of its development is an embryo to be regarded and treated as a person.Embryos could come to be viewed as a commodity, not as an embryo that could develop into a person.For therapeutic cloning, there is a question of whether it is right to create embryos for therapy, and destroy them in the process.A source of embryonic stem cells is unused embryos produced by in vitro fertilisation (IVF).Cultured stem cells could be contaminated with viruses which would be transferred to a patient.Mutations have been observed in stem cells cultured for a number of generations, and some mutated stem cells have been observed to behave like cancer cells.The cells would have to be collected before birth or shortly after - some clinics offer to store blood from the umbilical cord when a person is born. The difficulty in obtaining and storing a patient's embryonic stem cells.The current difficulty in finding suitable stem cell donors.There is no guarantee how successful these therapies will be.Some variables which would be considered when discussing stem cells include: Clinical issues Sometimes, there are no right or wrong answers, or even answers at all. It is important to obtain a balanced view. They will also depend on whether the stem cells are to be used for therapy or research. These issues will be different for growth and transplant of adult, embryonic and therapeutically-cloned stem cells. But there are clinical, ethical and social issues with their use. Stem cells have great potential, in treating patients with currently untreatable conditions, growing organs for transplants and research. Benefits and risks associated with the use of stem cells in medicine 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
